NA Labetalol Hydrochloride Impurity - Anax Laboratories
Por um escritor misterioso
Descrição
Anax Laboratories provides Chemical industry users with Impurities of Labetalol Hydrochloride Impurity(NA) Boiling point Melting point, Labetalol Hydrochloride Impurity (NA ) Density MSDS Formula Use,If You also need to Labetalol Hydrochloride Impurity (NA ) Other information,welcome to contact us.

Amneal Pharmaceuticals, Inc. - Amneal Pharmaceuticals LLC Issues Voluntary Nationwide Recall of Metformin Hydrochloride Extended Release Tablets, USP, 500 mg and 750 mg, due to Detection of N-Nitrosodimethylamine (NDMA) Impurity to Consumer Level
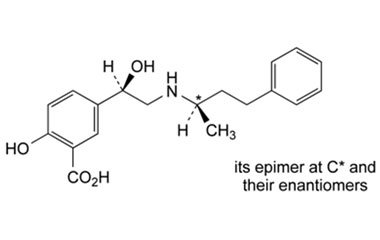
Labetalol Hydrochloride-impurities
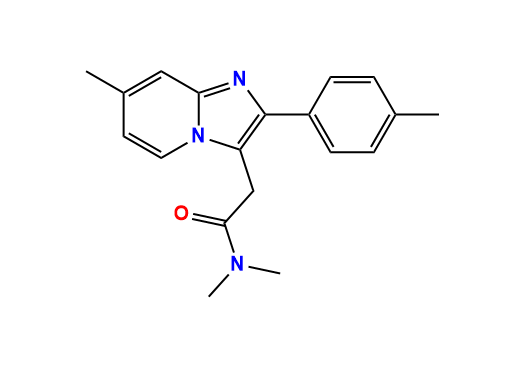
NA Zolpidem Impurity A - Anax Laboratories

PDF) Determination of alcuronium dichloride in plasma by high-performance liquid chromatography without solvent extraction
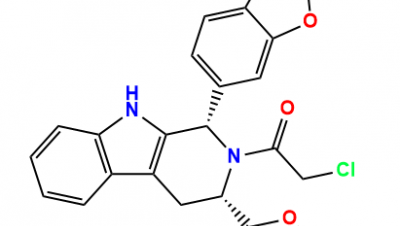
Impurities Index, CAS Number, Product Code

Structures of labetalol hydrochloride and its related impurity
Random Word Generator ― Perchance
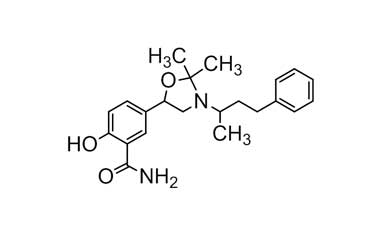
CAS No : 2445226-37-7, Product Name : Labetalol Hydrochloride - Impurity G (Hydrochloride Salt)

LABETALOL HYDROCHLORIDE INJECTION, USP

Sno Edited, PDF
Product Details Pfizer Hospital

New lonching - Integrity pharmaceutical

Anti-NALCN/VGCNL1 (extracellular) Antibody, #ASC-022
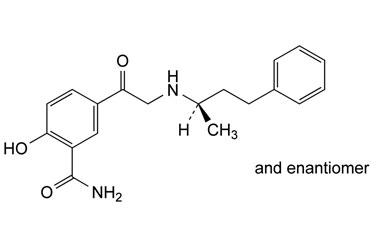
Labetalol Hydrochloride-impurities
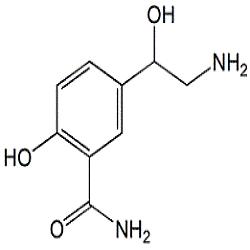
Labetalol Acetonide Impurity - Opulent Pharma
de
por adulto (o preço varia de acordo com o tamanho do grupo)